GROUP RESEARCH AREA
Research Topics:
- Photocatalytic Nitrogen Reduction Reaction
- Photocatalytic-Organic and Dye Pollutant Removal
- Supercapacitors
- Electrochemical Water splitting
- Electrochemical Sensor
- Urea Electrolysis
- Zinc-air battery
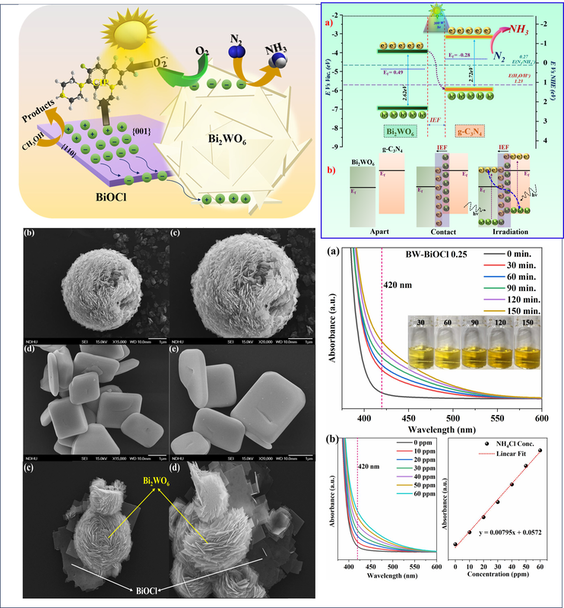
Photocatalytic Nitrogen Reduction Reaction
Ammonia, being the building block for various fertilizers like urea, diammonium phosphate (DAP) and pharmaceutical products, is an indispensable agricultural and industrial chemical. It is considered as a source for clean energy storage and potential hydrogen carrier. At present the industrial synthesis of ammonia solely depends on Haber-Bosch process. The high temperature and pressure requirement makes Haber-Bosch process the most energy intensive process and contributes to 1-2% of total global energy consumption. Photocatalytic nitrogen reduction is a promising green alternative to the traditional Haber-Bosch process, offering a sustainable, energy-efficient, and environmentally friendly method of producing ammonia. The process involves the use of photocatalysts to reduce nitrogen to ammonia at ambient temperature and pressure by using light as the source of energy and water as the source of proton.
Ammonia, being the building block for various fertilizers like urea, diammonium phosphate (DAP) and pharmaceutical products, is an indispensable agricultural and industrial chemical. It is considered as a source for clean energy storage and potential hydrogen carrier. At present the industrial synthesis of ammonia solely depends on Haber-Bosch process. The high temperature and pressure requirement makes Haber-Bosch process the most energy intensive process and contributes to 1-2% of total global energy consumption. Photocatalytic nitrogen reduction is a promising green alternative to the traditional Haber-Bosch process, offering a sustainable, energy-efficient, and environmentally friendly method of producing ammonia. The process involves the use of photocatalysts to reduce nitrogen to ammonia at ambient temperature and pressure by using light as the source of energy and water as the source of proton.
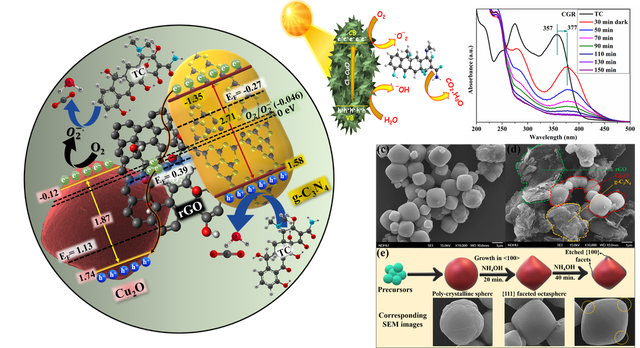
Photocatalytic-organic and dye pollutant removal
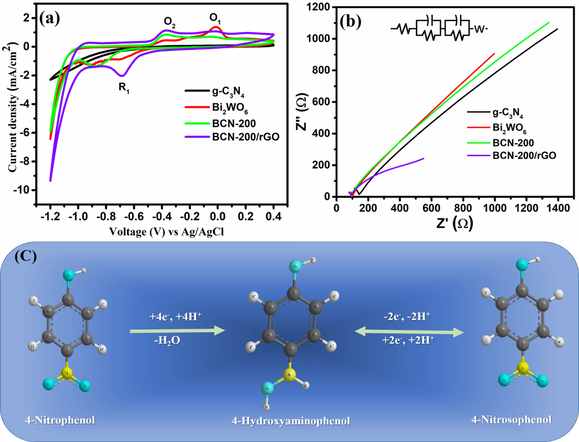
Photocatalytic water pollutant treatment are important research area for addressing global environmental issues. Treatment of wastewater containing persistent organic and inorganic pollutants, prior to disposal into the environment, has attracted substantial interest over the past decades. The project will focus on fabrication of low cost, non toxic and highly efficient photocatalysts and their application in wastewater treatment. The relevant reaction mechanism will also be investigated by spectroscopies and other advanced technologies, which will be fed back to the material synthesis to improve the catalytic efficiency.
Photocatalytic water pollutant treatment are important research area for addressing global environmental issues. Treatment of wastewater containing persistent organic and inorganic pollutants, prior to disposal into the environment, has attracted substantial interest over the past decades. The project will focus on fabrication of low cost, non toxic and highly efficient photocatalysts and their application in wastewater treatment. The relevant reaction mechanism will also be investigated by spectroscopies and other advanced technologies, which will be fed back to the material synthesis to improve the catalytic efficiency.
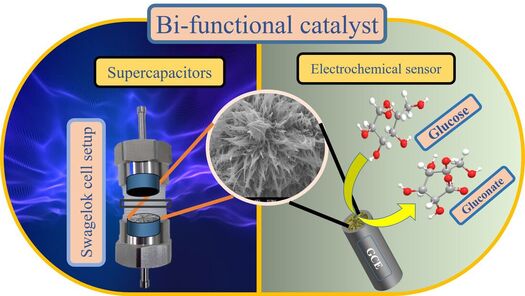
Supercapacitor
Supercapacitors also called electrochemical capacitors and ultra-capacitors, these energy storage devices can store and deliver energy at relatively high rates in comparison to batteries. It has a simple mechanism of energy storage of charge-separation at the electrochemical interface between the electrode and the electrolyte. Supercapacitors owe long cycle life and rapid charging and discharging at high power densities, due to these advantages it has found its applicability in load cranes, forklifts, electric vehicles, electric utilities, factory power backup and so on.
Supercapacitors also called electrochemical capacitors and ultra-capacitors, these energy storage devices can store and deliver energy at relatively high rates in comparison to batteries. It has a simple mechanism of energy storage of charge-separation at the electrochemical interface between the electrode and the electrolyte. Supercapacitors owe long cycle life and rapid charging and discharging at high power densities, due to these advantages it has found its applicability in load cranes, forklifts, electric vehicles, electric utilities, factory power backup and so on.
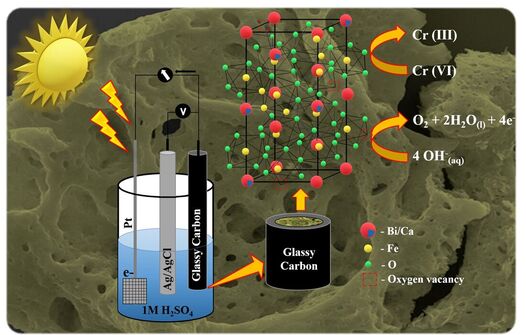
Electrochemical water splitting
To tackle carbon-based fuel, developing efficient electrocatalysts for energy conversion or energy storage systems like metal-air batteries, alkaline water electrolysis are current research trends. These systems consist of two electrodes of anode and cathode, where the anode part involve oxygen evolution reaction (OER), and the cathode part involve hydrogen evolution reaction (HER) or oxygen reduction reaction (ORR). The four electron-proton processes of OER requires higher overpotential, which hinders the practical application. To keep this in mind our research group develops inexpensive perovskite-based catalysts and studied their OER electro-catalytic activity. At the same time, we have also developed a Cu (copper) based electro-catalyst and analyze the HER activity.
To tackle carbon-based fuel, developing efficient electrocatalysts for energy conversion or energy storage systems like metal-air batteries, alkaline water electrolysis are current research trends. These systems consist of two electrodes of anode and cathode, where the anode part involve oxygen evolution reaction (OER), and the cathode part involve hydrogen evolution reaction (HER) or oxygen reduction reaction (ORR). The four electron-proton processes of OER requires higher overpotential, which hinders the practical application. To keep this in mind our research group develops inexpensive perovskite-based catalysts and studied their OER electro-catalytic activity. At the same time, we have also developed a Cu (copper) based electro-catalyst and analyze the HER activity.
Electrochemical sensors
Electrochemical sensors work according to electrochemical measuring principles, e.g. Amperometric, potentiometric or impedimetric. The electrochemical sensor properties such as sensitivity, selectivity and stability (reliability and durability) of sensors are mainly influenced by the measuring conditions respecting temperature, pressure and chemical environment. Electrochemical sensors are widely used in many branches of industry, environmental and medical monitoring, for investigation of metabolism and control of biological processes. They are well-established and powerful tools to gain real-time information for process control by in situ measurements of chemical composition without sampling.
Electrochemical sensors work according to electrochemical measuring principles, e.g. Amperometric, potentiometric or impedimetric. The electrochemical sensor properties such as sensitivity, selectivity and stability (reliability and durability) of sensors are mainly influenced by the measuring conditions respecting temperature, pressure and chemical environment. Electrochemical sensors are widely used in many branches of industry, environmental and medical monitoring, for investigation of metabolism and control of biological processes. They are well-established and powerful tools to gain real-time information for process control by in situ measurements of chemical composition without sampling.
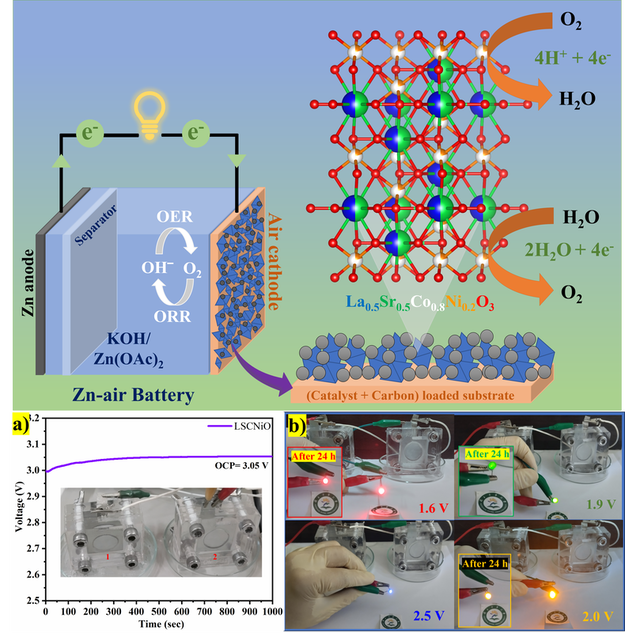
Zinc-air Battery
A zinc-air battery is a type of electrochemical battery that uses zinc and oxygen as the two active materials to generate electrical energy. These batteries belong to the class of metal-air batteries, where the anode is made of a metal (in this case, zinc) and the cathode is exposed to air, typically containing oxygen. Zinc-air batteries have gained attention due to their high energy density, low cost, and the abundance of zinc. The electrochemical combination of zinc, oxygen, and water to produce zinc hydroxide. The electrons released during the oxidation of zinc at the anode travel through an external circuit, providing electrical power.
One of the advantages of zinc-air batteries is their high energy density, which means they can store a large amount of energy for their size. This makes them attractive for applications such as electric vehicles and portable electronic devices. However, there are also challenges, including issues related to the stability of the cathode and the management of the electrolyte.
A zinc-air battery is a type of electrochemical battery that uses zinc and oxygen as the two active materials to generate electrical energy. These batteries belong to the class of metal-air batteries, where the anode is made of a metal (in this case, zinc) and the cathode is exposed to air, typically containing oxygen. Zinc-air batteries have gained attention due to their high energy density, low cost, and the abundance of zinc. The electrochemical combination of zinc, oxygen, and water to produce zinc hydroxide. The electrons released during the oxidation of zinc at the anode travel through an external circuit, providing electrical power.
One of the advantages of zinc-air batteries is their high energy density, which means they can store a large amount of energy for their size. This makes them attractive for applications such as electric vehicles and portable electronic devices. However, there are also challenges, including issues related to the stability of the cathode and the management of the electrolyte.